![Study on the Synthesis and Application of a Material 1-[2-Methyl-5-Phenyl-3-Thienyl]-2-[2-Methyl-5-(3-Aldehyde-4-Methoxy)-3-Thienyl] Perfluorocyclopentene | Scientific.Net Study on the Synthesis and Application of a Material 1-[2-Methyl-5-Phenyl-3-Thienyl]-2-[2-Methyl-5-(3-Aldehyde-4-Methoxy)-3-Thienyl] Perfluorocyclopentene | Scientific.Net](https://www.scientific.net/AMR.1142.85/preview.gif)
Study on the Synthesis and Application of a Material 1-[2-Methyl-5-Phenyl-3-Thienyl]-2-[2-Methyl-5-(3-Aldehyde-4-Methoxy)-3-Thienyl] Perfluorocyclopentene | Scientific.Net
The excited state antiaromatic benzene ring: a molecular Mr Hyde? - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C5CS00057B

Organocatalytic Atroposelective Aldol Condensation: Synthesis of Axially Chiral Biaryls by Arene Formation - Link - 2014 - Angewandte Chemie International Edition - Wiley Online Library

Bicyclic Phenyl–Ethynyl Architectures: Synthesis of a 1,4‐Bis(phenylbuta‐1,3‐diyn‐1‐yl) Benzene Banister - Bannwart - 2021 - Chemistry – A European Journal - Wiley Online Library

Figure 2 | The nomenclature of fused-ring arenes and heterocycles: a guide to an increasingly important dialect of organic chemistry | SpringerLink

Synthesis and Crystal Structure Studies on 3-(4-Fluorophenyl)-1,5-Bis(pyridyl)-1,5-Pentanedione | Scientific.Net

Common phenolic compounds in plants comprise an aromatic ring, bear one... | Download Scientific Diagram
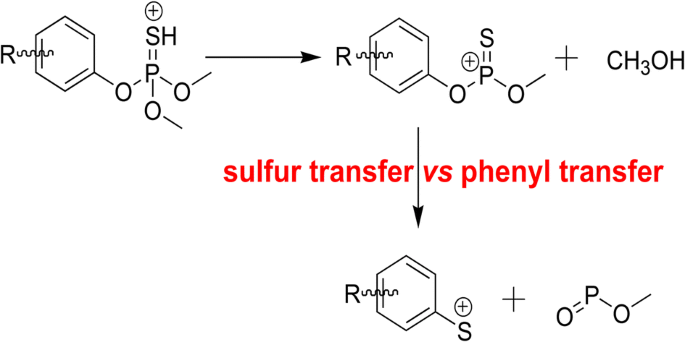
Sulfur Transfer Versus Phenyl Ring Transfer in the Gas Phase: Sequential Loss of CH3OH and CH3O–P=O from Protonated Phosphorothioates | SpringerLink

Aromatic Rings in Chemical and Biological Recognition: Energetics and Structures - Salonen - 2011 - Angewandte Chemie International Edition - Wiley Online Library