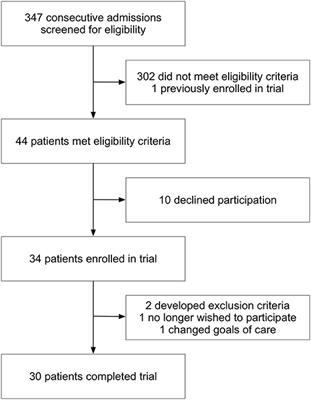
PLOS ONE: A Randomized Open-Label Trial of Artesunate- Sulfadoxine-Pyrimethamine with or without Primaquine for Elimination of Sub-Microscopic P. falciparum Parasitaemia and Gametocyte Carriage in Eastern Sudan

Single center, open label dose escalating trial evaluating once weekly oral ixazomib in ART-suppressed, HIV positive adults and effects on HIV reservoir size in vivo - eClinicalMedicine
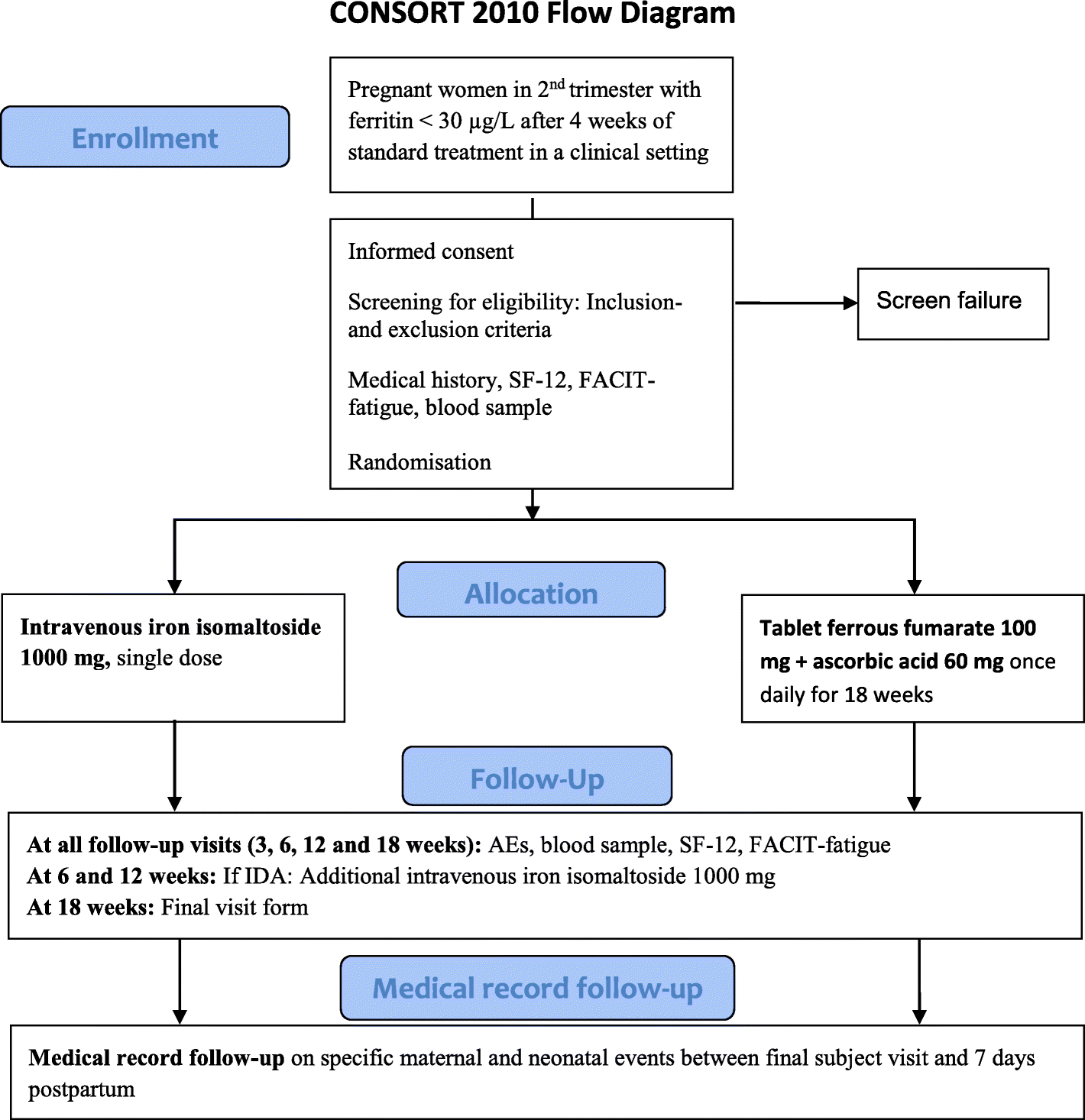
Intravenous iron isomaltoside versus oral iron supplementation for treatment of iron deficiency in pregnancy: protocol for a randomised, comparative, open-label trial | Trials | Full Text
![PDF] Open-label versus double-blind placebo treatment in irritable bowel syndrome: study protocol for a randomized controlled trial | Semantic Scholar PDF] Open-label versus double-blind placebo treatment in irritable bowel syndrome: study protocol for a randomized controlled trial | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/60afa7902fddea770036d6744ee84027545bd238/3-Figure1-1.png)
PDF] Open-label versus double-blind placebo treatment in irritable bowel syndrome: study protocol for a randomized controlled trial | Semantic Scholar

Phase I/II open-label trial of intravenous allogeneic mesenchymal stromal cell therapy in adults with recessive dystrophic epidermolysis bullosa - Journal of the American Academy of Dermatology

Safety and tolerability of adjunctive lacosamide in a pediatric population with focal seizures – An open-label trial - Seizure - European Journal of Epilepsy

Understanding Clinical Trial Terminology: What is an Open Label Clinical Trial? - Concert Pharmaceuticals

Effects of antiplatelet therapy on stroke risk by brain imaging features of intracerebral haemorrhage and cerebral small vessel diseases: subgroup analyses of the RESTART randomised, open-label trial - The Lancet Neurology
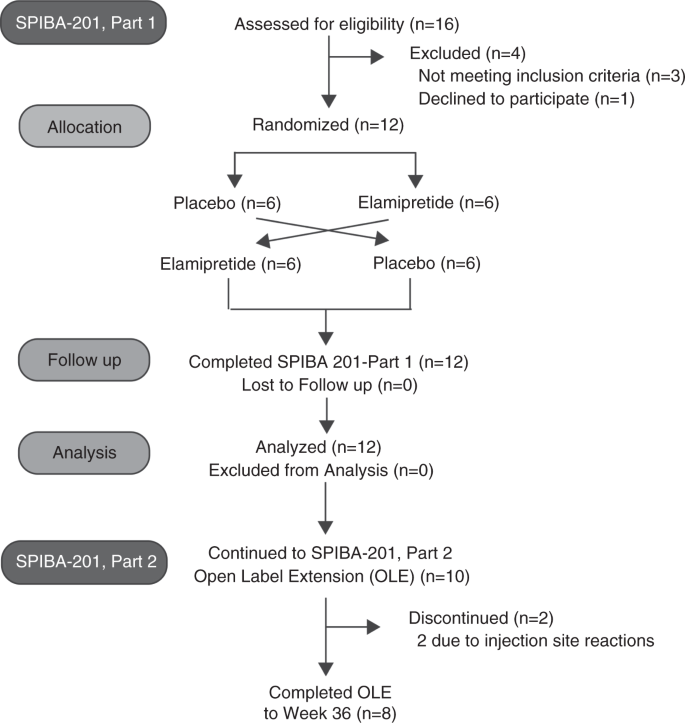
A phase 2/3 randomized clinical trial followed by an open-label extension to evaluate the effectiveness of elamipretide in Barth syndrome, a genetic disorder of mitochondrial cardiolipin metabolism | Genetics in Medicine

Phase II single arm open label multicentre clinical trial to evaluate the efficacy and side effects of a combination of gefitinib and methotrexate to treat tubal ectopic pregnancies (GEM II): study protocol