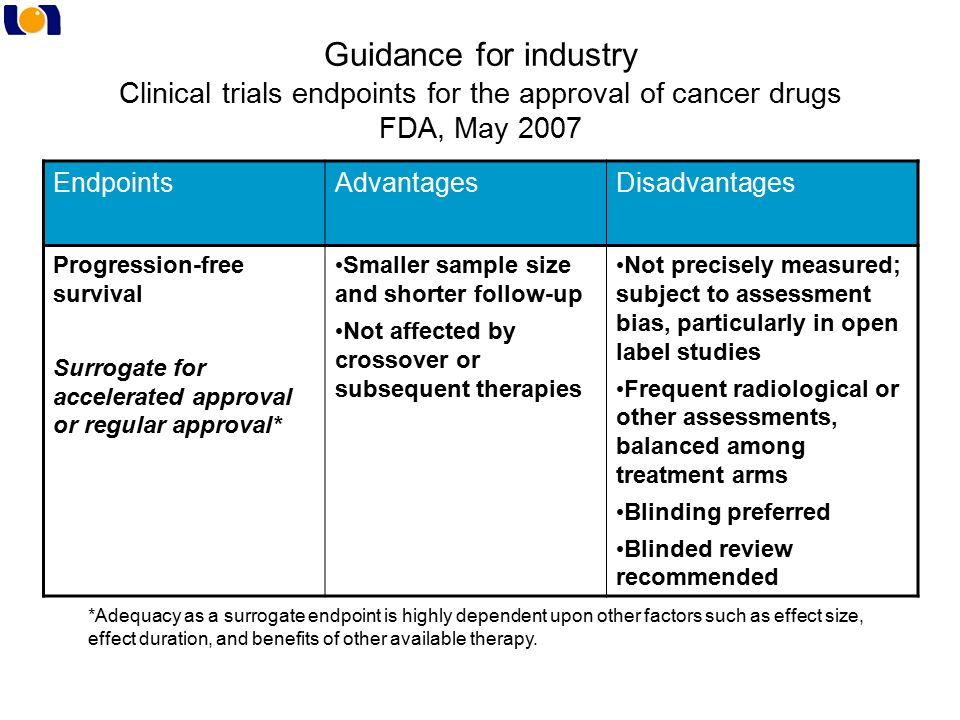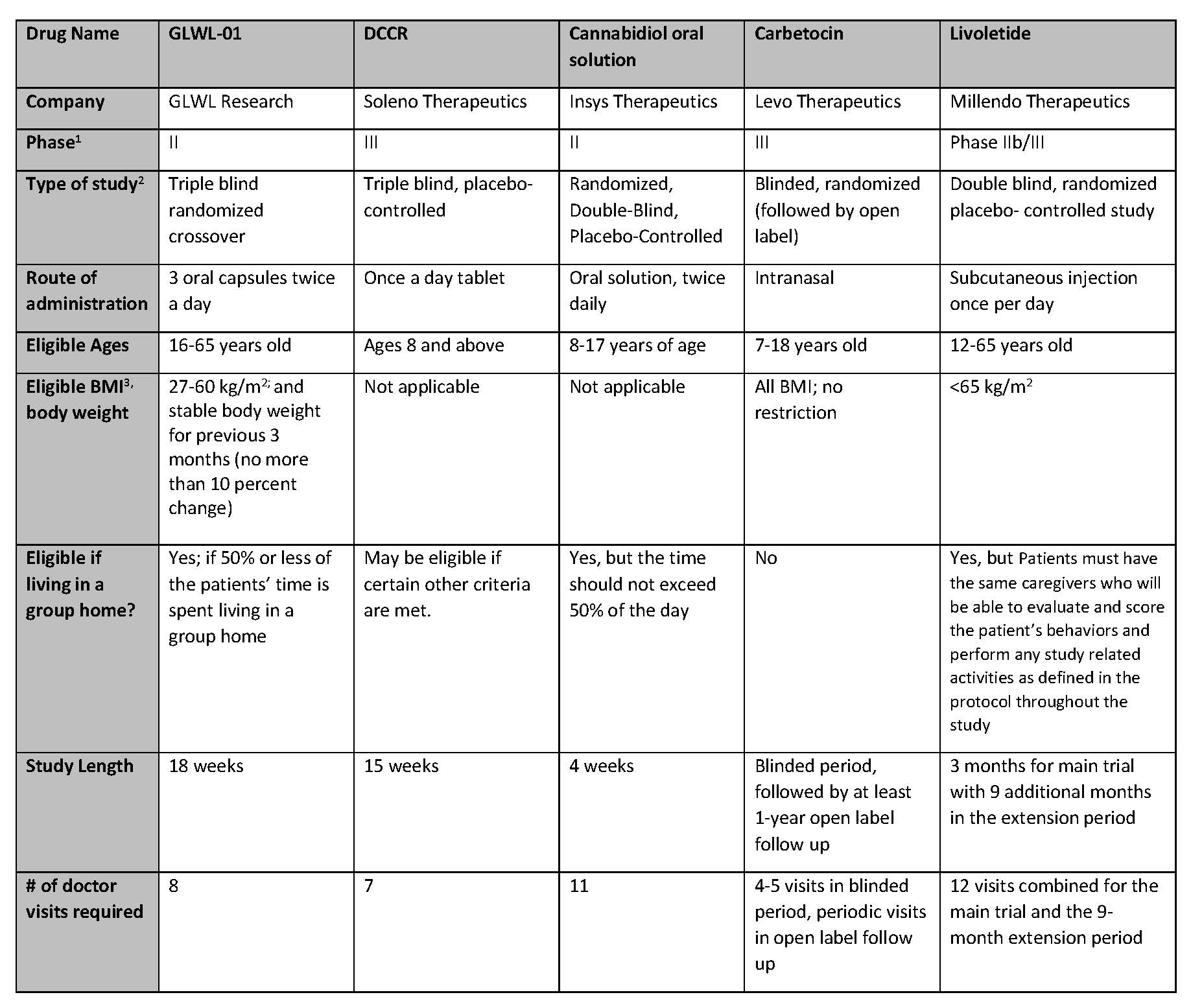
Summary Of Active Clinical Trials For Prader-Willi Syndrome Hyperphagia - Prader-Willi Syndrome Association | USA

A Prospective, Randomized, Open-Label, Blinded, Endpoint Study Exploring Platelet Response to Half-Dose Prasugrel and Ticagrelor in Patients with the Acute Coronary Syndrome: HOPE-TAILOR Study | Semantic Scholar

Alteplase versus tenecteplase for thrombolysis after ischaemic stroke (ATTEST): a phase 2, randomised, open-label, blinded endpoint study - The Lancet Neurology
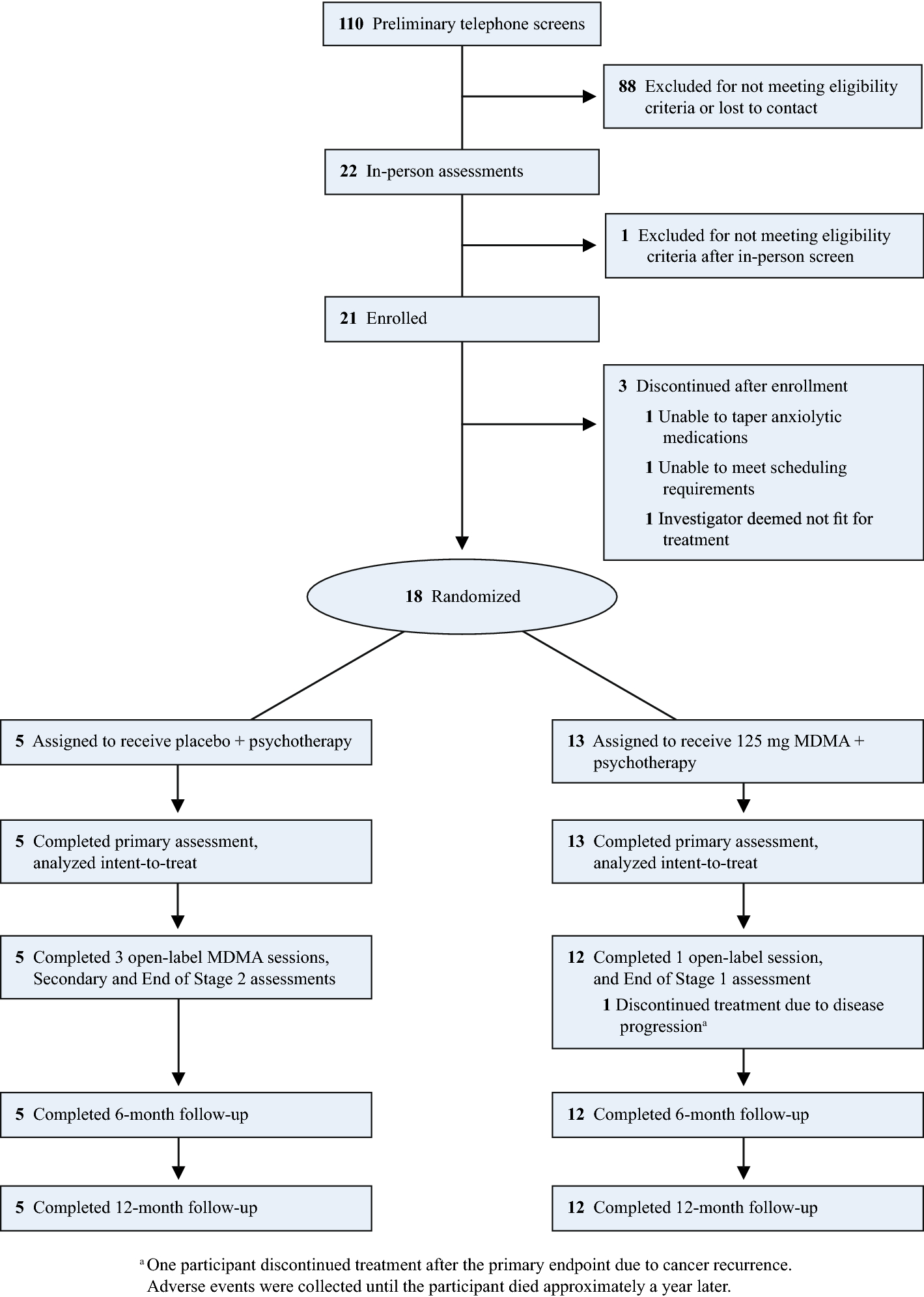
MDMA-assisted psychotherapy for treatment of anxiety and other psychological distress related to life-threatening illnesses: a randomized pilot study | Scientific Reports

A randomized, multicenter, open-label, blinded end point trial comparing the effects of spironolactone to chlorthalidone on left ventricular mass in patients with early-stage chronic kidney disease: Rationale and design of the SPIRO-CKD
PLOS Medicine: Preventing microalbuminuria with benazepril, valsartan, and benazepril–valsartan combination therapy in diabetic patients with high-normal albuminuria: A prospective, randomized, open-label, blinded endpoint (PROBE) study

1 SECOND AUSTRALIAN NATIONAL BLOOD PRESSURE STUDY (ANBP-2) Enalapril/ACEI vs. HCTZ, n = 6,083 Randomized, open-label (blinded endpoint review) All CV events. - ppt download

A multicenter, randomized, placebo-controlled, double-blind phase 3 trial with open-arm comparison indicates safety and efficacy of nephroprotective therapy with ramipril in children with Alport's syndrome - ScienceDirect

Methods of a large prospective, randomised, open-label, blinded end-point study comparing morning versus evening dosing in hypertensive patients: the Treatment In Morning versus Evening (TIME) study | BMJ Open
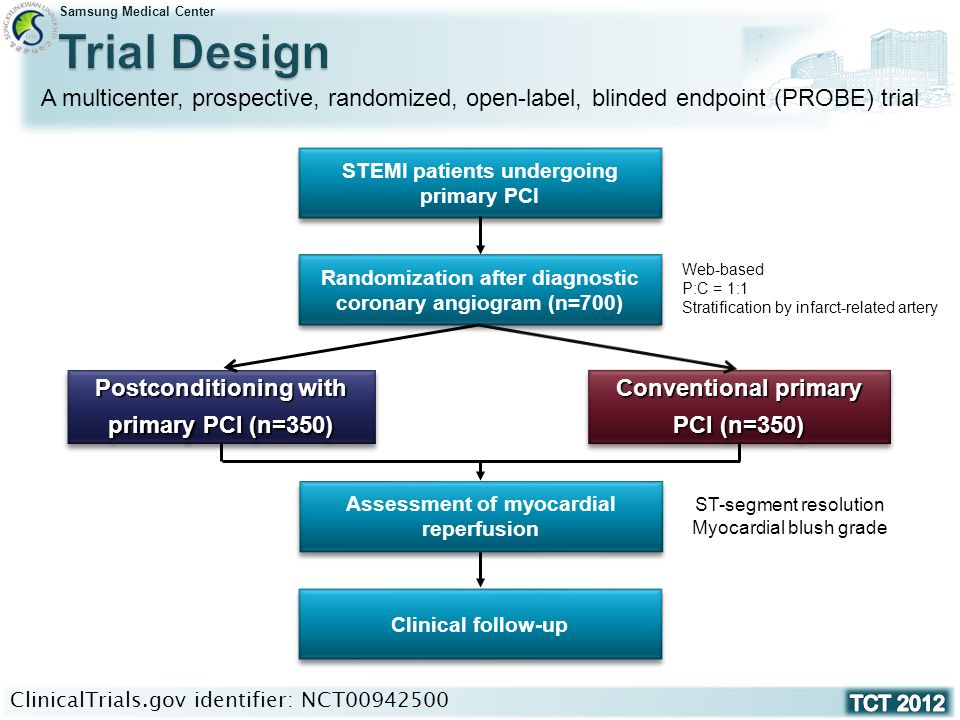
Effect of Postconditioning on Myocardial Reperfusion during Primary Percutaneous Coronary Intervention Joo-Yong Hahn / Hyeon-Cheol Gwon On behalf of the. - ppt video online download

A multicenter, randomized, placebo-controlled, double-blind phase 3 trial with open-arm comparison indicates safety and efficacy of nephroprotective therapy with ramipril in children with Alport's syndrome - ScienceDirect
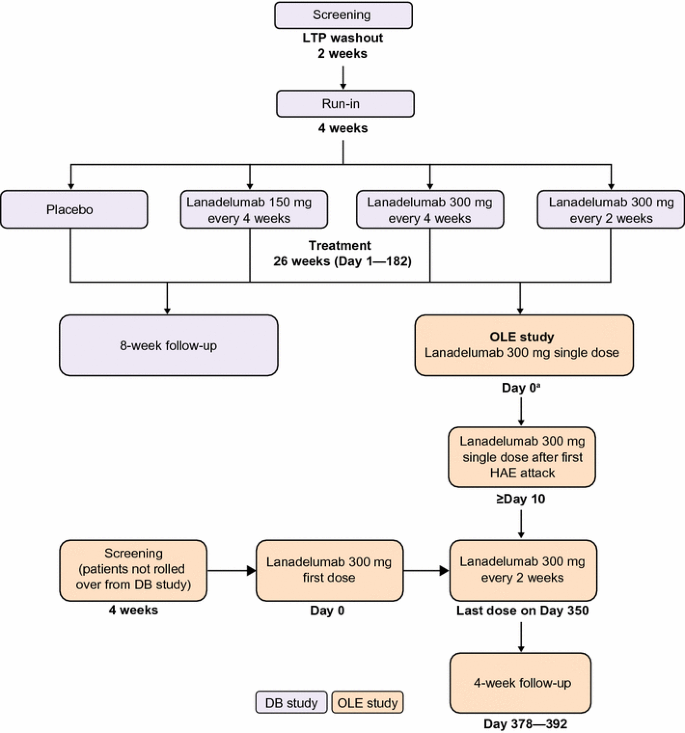
An open-label study to evaluate the long-term safety and efficacy of lanadelumab for prevention of attacks in hereditary angioedema: design of the HELP study extension | Clinical and Translational Allergy | Full
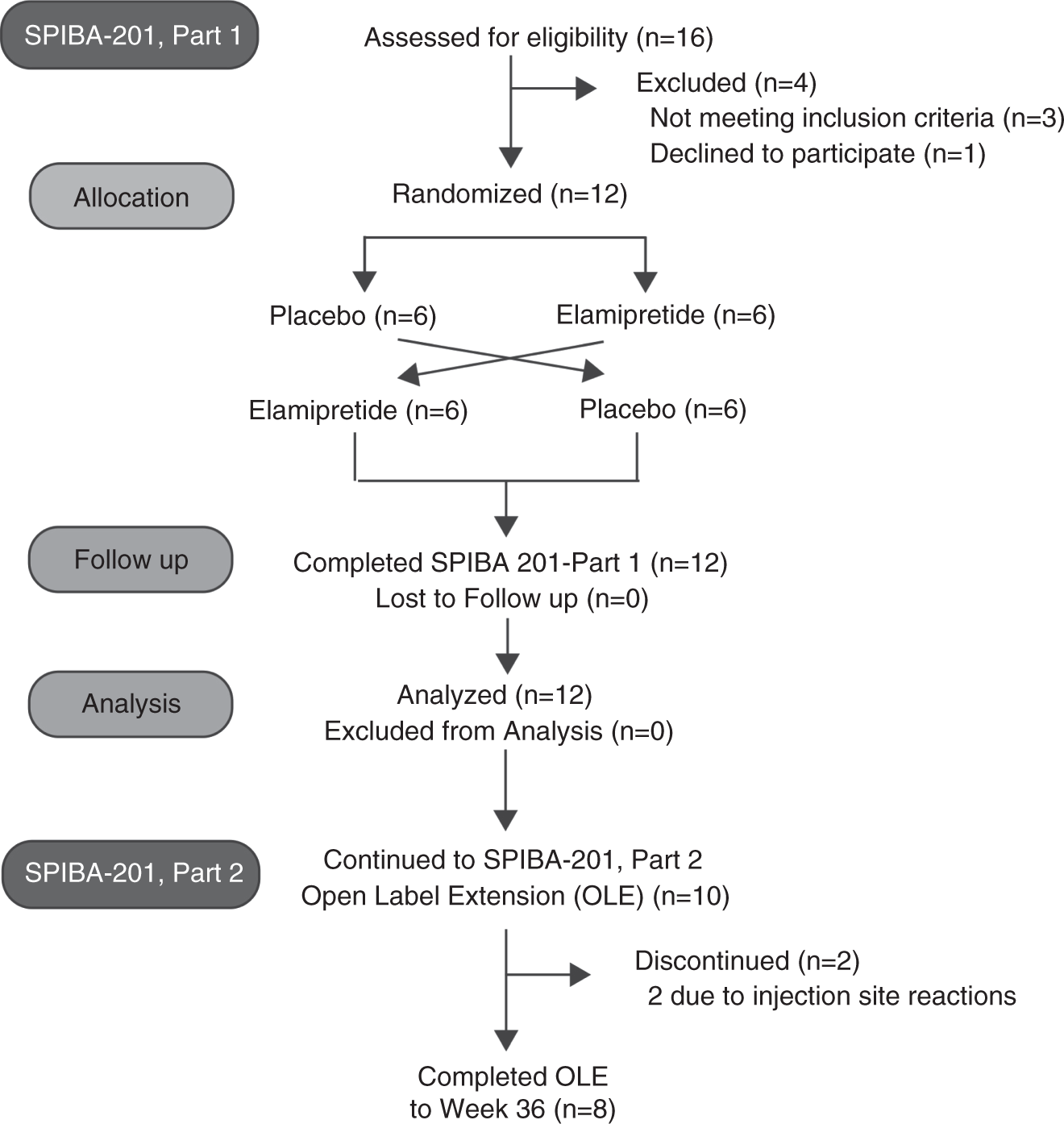
A phase 2/3 randomized clinical trial followed by an open-label extension to evaluate the effectiveness of elamipretide in Barth syndrome, a genetic disorder of mitochondrial cardiolipin metabolism | Genetics in Medicine

Methodology of a large prospective, randomised, open, blinded endpoint streamlined safety study of celecoxib versus traditional non-steroidal anti-inflammatory drugs in patients with osteoarthritis or rheumatoid arthritis: protocol of the standard care ...

Comparing the effects of ipragliflozin versus metformin on visceral fat reduction and metabolic dysfunction in Japanese patients with type 2 diabetes treated with sitagliptin: A prospective, multicentre, open‐label, blinded‐endpoint, randomized ...