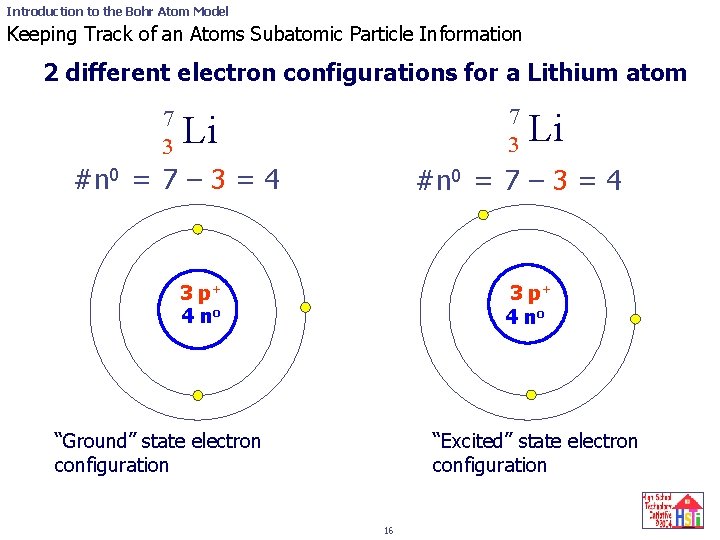
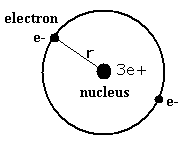
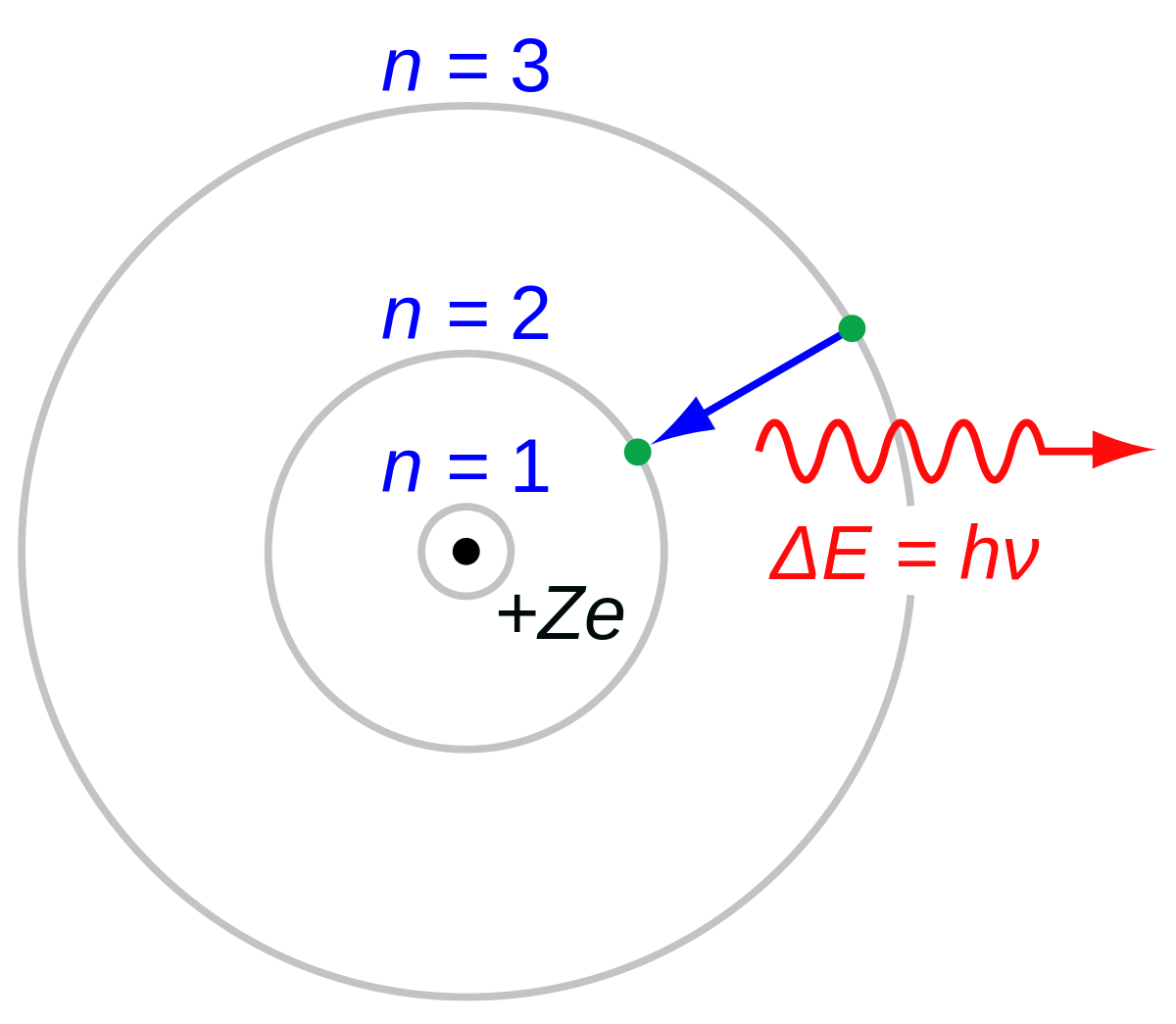
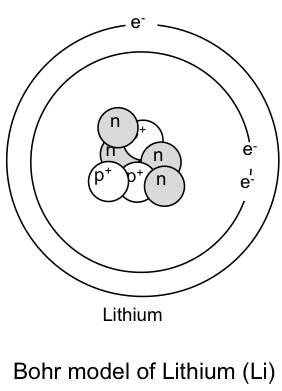
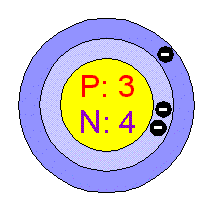
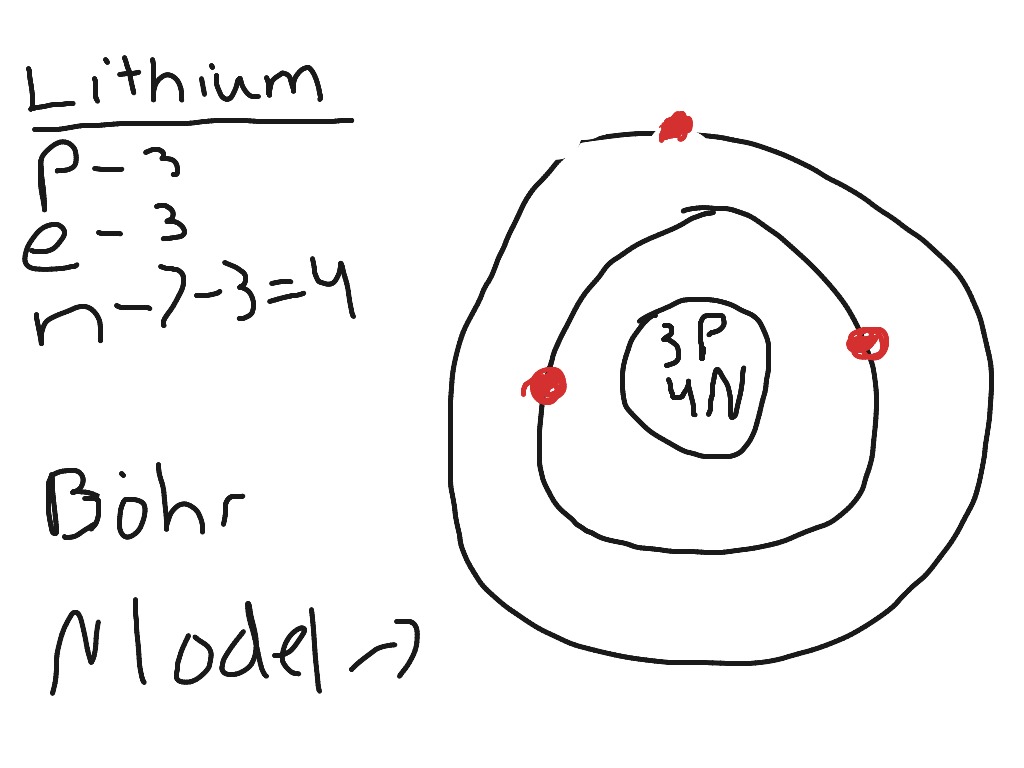
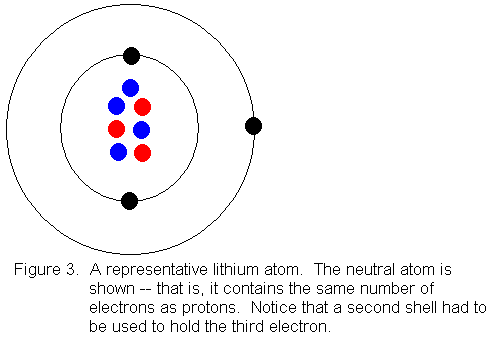
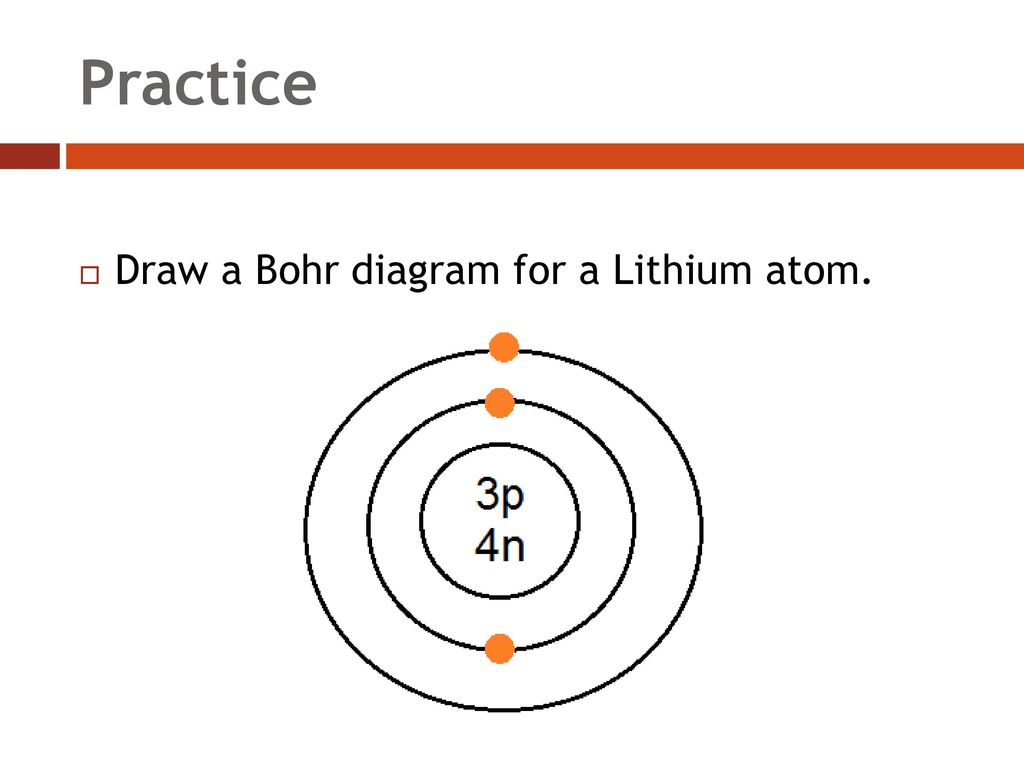
The following is a Bohr Model representation of a Lithium Atom:The first ionization energy for the lithium atom is 520.kJ/mol What is the wavelength (in nm) associated with the first ionization energy?

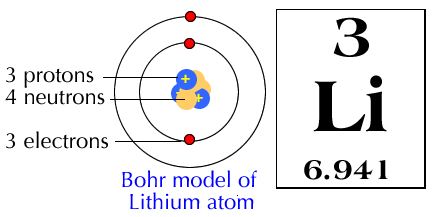
Lithium atom Bohr model Atomic number Particle, chemical atom, chemical Element, symmetry png | PNGEgg
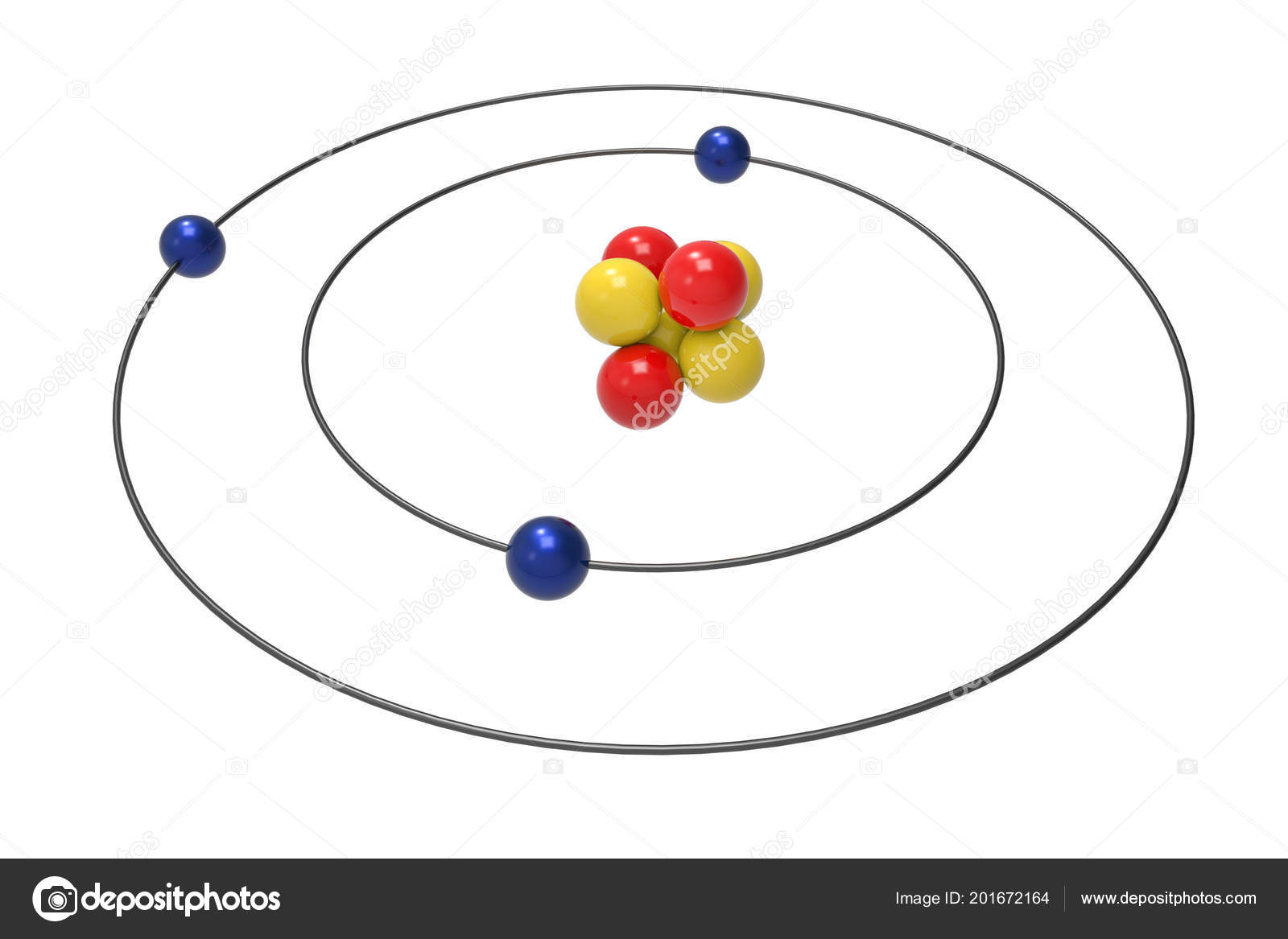
Bohr Model Lithium Atom Proton Neutron Electron Science Chemical Concept Stock Photo by ©Ema88 201672164