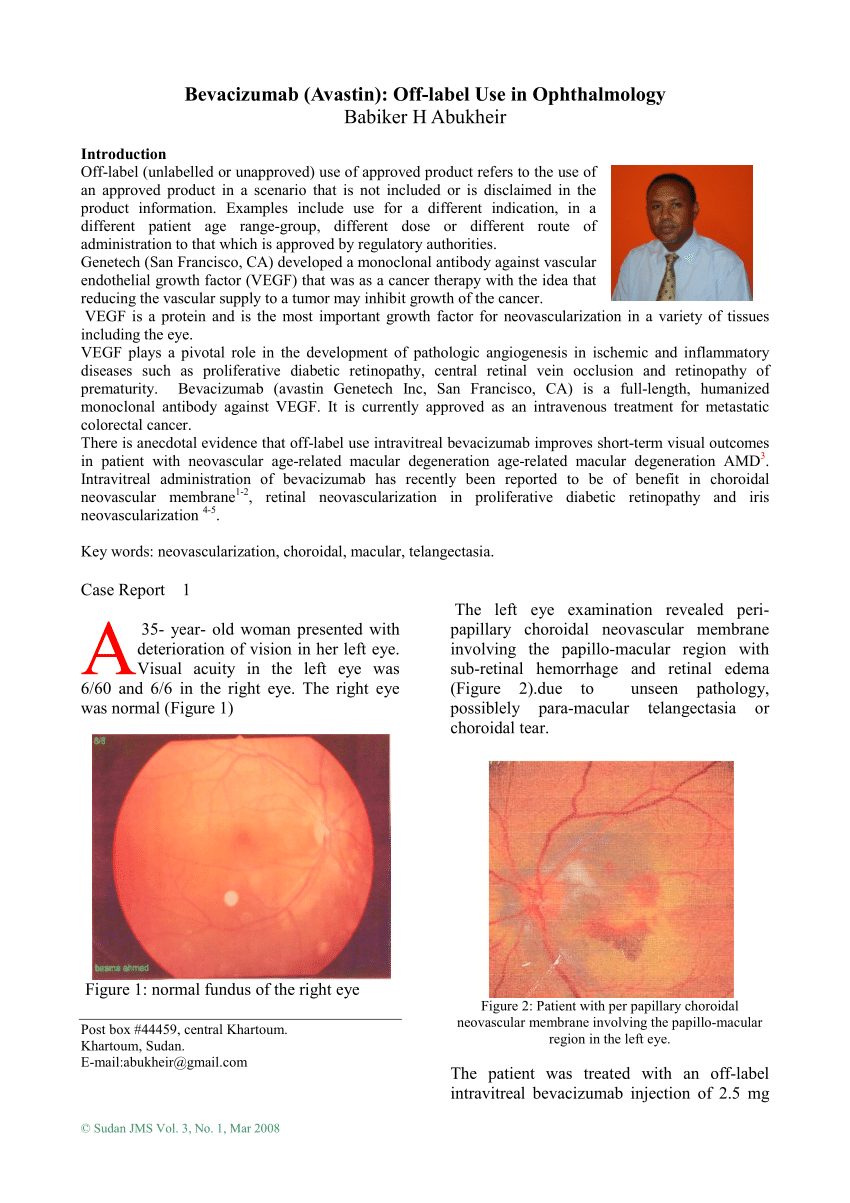
Bevacizumab biosimilar BEVZ92 versus reference bevacizumab in combination with FOLFOX or FOLFIRI as first-line treatment for metastatic colorectal cancer: a multicentre, open-label, randomised controlled trial - The Lancet Gastroenterology & Hepatology
PLOS ONE: A Safety Review and Meta-Analyses of Bevacizumab and Ranibizumab: Off-Label versus Goldstandard
Genentech: Statements | Genentech Statement on Counterfeit Drug Labeled as Avastin® (Bevacizumab) in the United States

ICD-9/10 codes to define serious adverse events for Bevacizumab based... | Download Scientific Diagram

Combination therapy of bevacizumab with either S-1 and irinotecan or mFOLFOX6/CapeOX as first-line treatment of metastatic colorectal cancer (TRICOLORE): Exploratory analysis of RAS status and primary tumour location in a randomised, open-label,
![PDF] Off-label use of bevacizumab for wet age-related macular degeneration in Europe | Semantic Scholar PDF] Off-label use of bevacizumab for wet age-related macular degeneration in Europe | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/dd8a688b93d7c74f55bbed645b027d6775de792f/6-Table1-1.png)
PDF] Off-label use of bevacizumab for wet age-related macular degeneration in Europe | Semantic Scholar

BRIDGE: An Open-Label Phase II Trial Evaluating the Safety of Bevacizumab + Carboplatin/Paclitaxel as First-Line Treatment for Patients with Advanced, Previously Untreated, Squamous Non-small Cell Lung Cancer - Journal of Thoracic Oncology
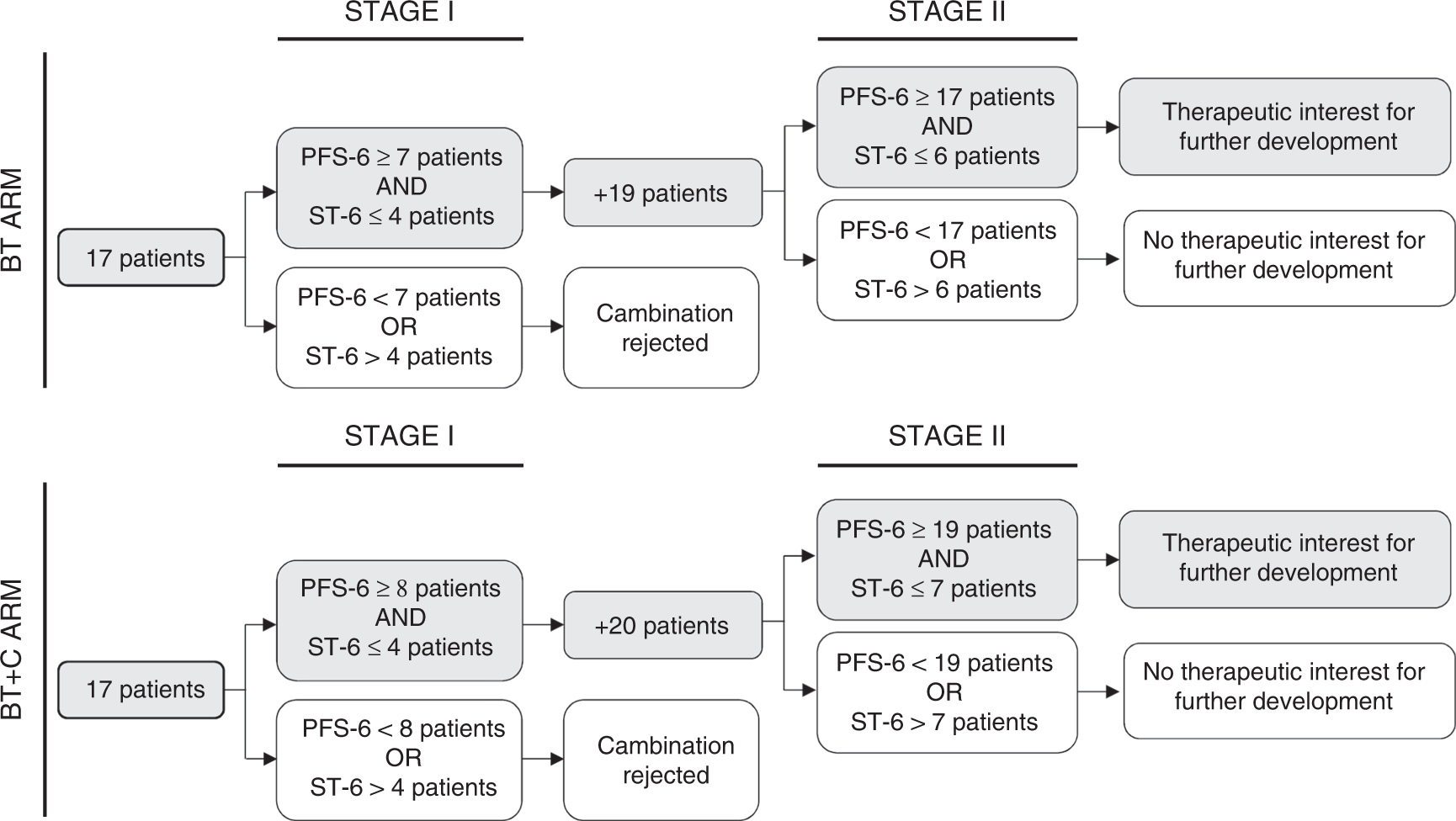
Multicenter, randomised, open-label, non-comparative phase 2 trial on the efficacy and safety of the combination of bevacizumab and trabectedin with or without carboplatin in women with partially platinum-sensitive recurrent ovarian cancer

TAS-102 with or without bevacizumab in patients with chemorefractory metastatic colorectal cancer: an investigator-initiated, open-label, randomised, phase 2 trial - The Lancet Oncology

Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open- label, phase 3 trial - The Lancet

Bevacizumab plus capecitabine versus capecitabine alone in elderly patients with previously untreated metastatic colorectal cancer (AVEX): an open-label, randomised phase 3 trial - The Lancet Oncology